A newly published workshop report in Vaccine calls for increased global collaboration and investment to accelerate the development and implementation of vaccines to prevent malaria in pregnancy (MiP), a major yet under-recognized public health challenge.
The publication summarizes key outcomes from a multistakeholder workshop held under the ADVANCE-VAC4PM project and co-organised by the European Vaccine Initiative and La Fondation pour la Recherche Scientifique in June 2025. The event brought together researchers, regulators, policymakers, civil society representatives, and global health experts to define strategies for MiP vaccine development and deployment.
Malaria continues to impose a significant burden, particularly in sub-Saharan Africa, where pregnant women are disproportionately affected. Placental malaria can lead to severe health outcomes for mothers and newborns, with the highest risk observed in first and second pregnancies. Existing malaria control measures remain insufficient to fully protect pregnant women. Vaccines specifically targeting MiP could complement current interventions by inducing immunity similar to that naturally acquired over multiple pregnancies. To achieve maximum impact, such vaccines would need to provide long-lasting protection and be administered before a woman’s first pregnancy.
The workshop integrated scientific, regulatory, cost-effectiveness, and socio-behavioural perspectives, providing a platform for coordinated dialogue among global stakeholders on development, evaluation, and implementation strategies.
Key recommendations include strengthening communication and community engagement, defining appropriate clinical trial endpoints, reinforcing safety monitoring systems, and ensuring early regulatory alignment.
“Malaria in pregnancy remains a significant yet under-recognized threat to maternal and newborn health,” said Flavia D'Alessio, Project Coordinator and Head of Vaccine Research at the European Vaccine Initiative. “The report underscores the urgent need to accelerate the development of targeted vaccines and to ensure that the necessary systems are in place to support and promote equally their safe and effective use”.
The report concludes with a strong call for sustained international collaboration and increased national investment to ensure that MiP vaccines become a viable and effective component of global malaria prevention strategies.
Read full article: https://doi.org/10.1016/j.vaccine.2026.128505
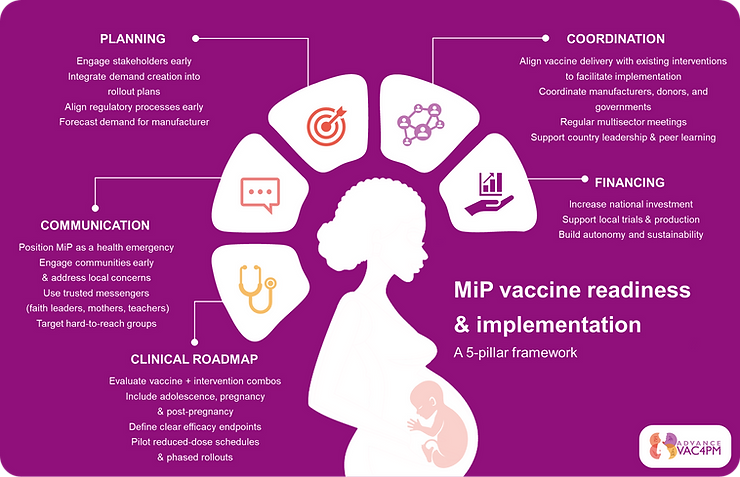
Malaria in pregnancy vaccine readiness and implementation framework based on the key workshop recommendations.

Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or the European Health and Digital Executive Agency. Neither the European Union nor the granting authority can be held responsible for them.
---------------------------------------
This article is republished under the authorization of the European Vaccine Initiative (EVI), and is for educational and exchange purposes only. Original link: https://www.euvaccine.eu/post/a-roadmap-for-malaria-in-pregnancy-vaccines-advance-vac4pm-workshop-findings-now-published. All copyrights belong to the original author.





