The European Vaccine Initiative (EVI) is supporting the clinical development of a novel Nipah virus vaccine candidate developed by the University of Tokyo, Japan.
February 2026
The vaccine is based on a recombinant measles virus vector incorporating genetic information from the Nipah virus. Preclinical studies have demonstrated its safety and protective efficacy in animal models.
The first-in-human clinical trial is scheduled to begin in April in Belgium, with EVI collaborating closely with the University of Tokyo to support the transition from preclinical research to clinical evaluation. EVI is contributing its expertise in clinical trial coordination, regulatory processes, and international partnerships.
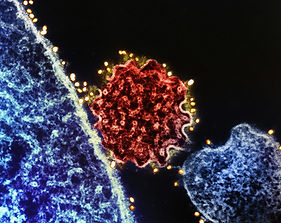
Colorized transmission electron micrograph of a single Nipah virus particle (center, colorized red) that has budded from the surface of an infected cell (blue). Credit: NIAD
Following the initial trial in Europe, Phase 1/2 clinical trials are planned to be conducted in Bangladesh, a country with previous Nipah virus outbreaks. These studies will further assess the vaccine’s safety and immunogenicity in both adults and children. Funding for early-stage clinical development is being provided by Japan’s Strategic Center for Advanced Research and Development (SCARDA).
In recent weeks, health authorities in India’s state of West Bengal have confirmed cases of Nipah virus infection amid ongoing surveillance and containment efforts. The Nipah virus is known for its high case‑fatality rate, typically estimated between 40 % and 75 % in documented outbreaks, and has historically caused significant mortality in South Asia in the absence of licensed vaccines or specific treatments.
Nipah virus is listed by the World Health Organization as a priority pathogen due to its high case‑fatality rate and epidemic potential. The recent isolated cases reported in South Asia underscore the ongoing need for preparedness and the development of effective preventive tools.
Through this collaboration, EVI reinforces its commitment to advancing vaccines against emerging infectious diseases with epidemic potential, supporting global health preparedness efforts.
Photo by National Institute of Allergy and Infectious Diseases on Unsplash
--------------------------------------------
This article is republished under the authorization of the European Vaccine Initiative (EVI), and is for educational and exchange purposes only. Original link:https://www.euvaccine.eu/post/evi-supports-first-in-human-clinical-trial-of-novel-nipah-virus-vaccine. All copyrights belong to the original author.





